Ema Templates
Ema Templates - Find information for clinical trial sponsors on how to log in or register for the clinical trials information system (ctis). Briefing document template for parallel hta coordination group (htacg)/european medicines agency (ema) joint scientific consultation (jsc) for medicinal products (mp) page contents details This project is funded by eu4health. Download the main template in your languages (i keep them on my second monitor when i’m translating). Read the qrd guidance documents on formatting, style and terminology. The european medicines agency's (ema) working group on quality review of documents (qrd) develops, reviews and updates templates for product information for use by applicants and marketing authorisation holders for human medicines. Volume 10 of the publication the rules governing medicinal products in the european union contains guidance documents applying to clinical trials. A number of documents in volume 10 have been revised and updated to bring them in line with the changes required by the clinical trials regulation (eu) no 536/2014. The committee for medicinal products for human use (chmp) and committee on advanced therapies (cat) should use the assessment report templates and documents listed below for the assessment of any new application in the centralised procedure. Ema, national competent authorities and the european commission are conducting an electronic product information (epi) pilot project to pilot use of the eu epi common standard in regulatory procedures. The european medicines agency's (ema) working group on quality review of documents (qrd) develops, reviews and updates templates for product information for use by applicants and marketing authorisation holders for human medicines. This project is funded by eu4health. Find information for clinical trial sponsors on how to log in or register for the clinical trials information system (ctis). Briefing document template for parallel hta coordination group (htacg)/european medicines agency (ema) joint scientific consultation (jsc) for medicinal products (mp) page contents details Volume 10 of the publication the rules governing medicinal products in the european union contains guidance documents applying to clinical trials. Download the main template in your languages (i keep them on my second monitor when i’m translating). From 1 january 2019, the formatted letter template will not be maintained by ema and the document and references will be removed from ema corporate website. Ema, national competent authorities and the european commission are conducting an electronic product information (epi) pilot project to pilot use of the eu epi common standard in regulatory procedures. Read the qrd guidance documents on formatting, style and terminology. A number of documents in volume 10 have been revised and updated to bring them in line with the changes required by the clinical trials regulation (eu) no 536/2014. Ema, national competent authorities and the european commission are conducting an electronic product information (epi) pilot project to pilot use of the eu epi common standard in regulatory procedures. Find information for clinical trial sponsors on how to log in or register for the clinical trials information system (ctis). Read the qrd guidance documents on formatting, style and terminology. The. From 1 january 2019, the formatted letter template will not be maintained by ema and the document and references will be removed from ema corporate website. Briefing document template for parallel hta coordination group (htacg)/european medicines agency (ema) joint scientific consultation (jsc) for medicinal products (mp) page contents details The european medicines agency's (ema) working group on quality review of. Read the qrd guidance documents on formatting, style and terminology. Briefing document template for parallel hta coordination group (htacg)/european medicines agency (ema) joint scientific consultation (jsc) for medicinal products (mp) page contents details Find information for clinical trial sponsors on how to log in or register for the clinical trials information system (ctis). Ema, national competent authorities and the european. Briefing document template for parallel hta coordination group (htacg)/european medicines agency (ema) joint scientific consultation (jsc) for medicinal products (mp) page contents details Read the qrd guidance documents on formatting, style and terminology. A number of documents in volume 10 have been revised and updated to bring them in line with the changes required by the clinical trials regulation (eu). A number of documents in volume 10 have been revised and updated to bring them in line with the changes required by the clinical trials regulation (eu) no 536/2014. From 1 january 2019, the formatted letter template will not be maintained by ema and the document and references will be removed from ema corporate website. The committee for medicinal products. From 1 january 2019, the formatted letter template will not be maintained by ema and the document and references will be removed from ema corporate website. This project is funded by eu4health. Find information for clinical trial sponsors on how to log in or register for the clinical trials information system (ctis). The european medicines agency's (ema) working group on. The european medicines agency's (ema) working group on quality review of documents (qrd) develops, reviews and updates templates for product information for use by applicants and marketing authorisation holders for human medicines. Read the qrd guidance documents on formatting, style and terminology. This project is funded by eu4health. Download the main template in your languages (i keep them on my. The committee for medicinal products for human use (chmp) and committee on advanced therapies (cat) should use the assessment report templates and documents listed below for the assessment of any new application in the centralised procedure. From 1 january 2019, the formatted letter template will not be maintained by ema and the document and references will be removed from ema. A number of documents in volume 10 have been revised and updated to bring them in line with the changes required by the clinical trials regulation (eu) no 536/2014. The committee for medicinal products for human use (chmp) and committee on advanced therapies (cat) should use the assessment report templates and documents listed below for the assessment of any new. Find information for clinical trial sponsors on how to log in or register for the clinical trials information system (ctis). Volume 10 of the publication the rules governing medicinal products in the european union contains guidance documents applying to clinical trials. Download the main template in your languages (i keep them on my second monitor when i’m translating). A number. Briefing document template for parallel hta coordination group (htacg)/european medicines agency (ema) joint scientific consultation (jsc) for medicinal products (mp) page contents details A number of documents in volume 10 have been revised and updated to bring them in line with the changes required by the clinical trials regulation (eu) no 536/2014. The european medicines agency's (ema) working group on quality review of documents (qrd) develops, reviews and updates templates for product information for use by applicants and marketing authorisation holders for human medicines. Read the qrd guidance documents on formatting, style and terminology. From 1 january 2019, the formatted letter template will not be maintained by ema and the document and references will be removed from ema corporate website. Find information for clinical trial sponsors on how to log in or register for the clinical trials information system (ctis). Ema, national competent authorities and the european commission are conducting an electronic product information (epi) pilot project to pilot use of the eu epi common standard in regulatory procedures. Download the main template in your languages (i keep them on my second monitor when i’m translating).EMA Example Audit Report Template 11 January 2022 PDF Audit
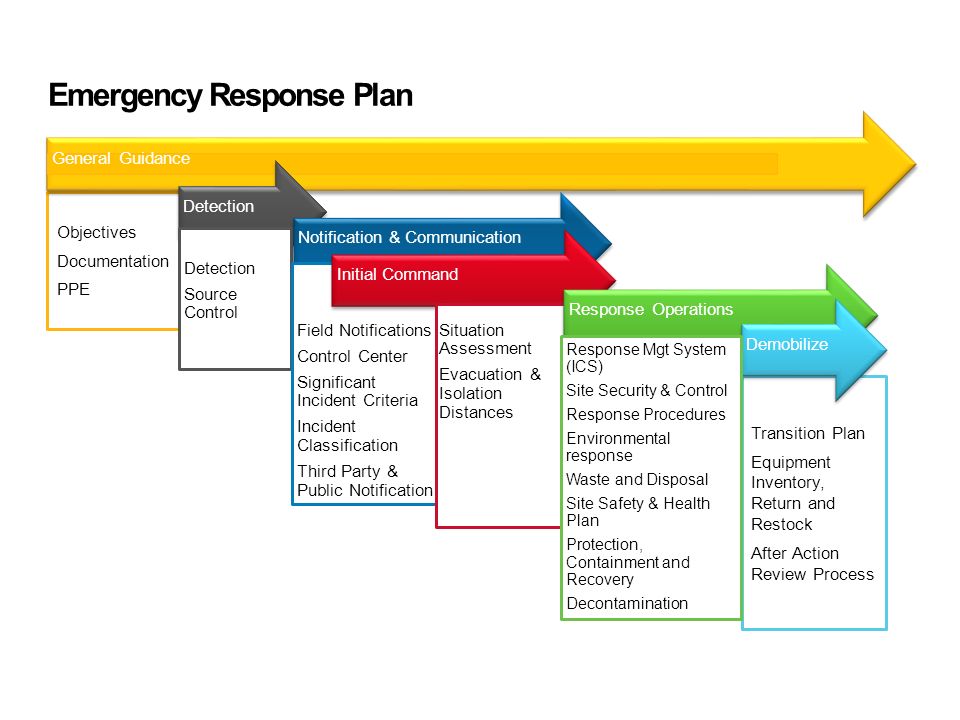
Emergency Management Plan 7+ Examples, Format, Pdf
8+ Emergency Management Plan Word Templates Free Downloads
Emergency Management Plan 7+ Examples, Format, Pdf
Ema Product Information Templates
Blank Ema Template + Colouringin Dragon Cutouts Supplies For Sensei
Ema Product Information Templates
Emergency Management Plan Template prntbl.concejomunicipaldechinu.gov.co
Incident Management Plan Template
Emergency Preparedness Plan Example Template Venngage
Volume 10 Of The Publication The Rules Governing Medicinal Products In The European Union Contains Guidance Documents Applying To Clinical Trials.
This Project Is Funded By Eu4Health.
The Committee For Medicinal Products For Human Use (Chmp) And Committee On Advanced Therapies (Cat) Should Use The Assessment Report Templates And Documents Listed Below For The Assessment Of Any New Application In The Centralised Procedure.
Related Post: